Near-infrared spectroscopy is used in astronomy for studying the atmospheres of cool stars where molecules can form. Typical applications of NIR spectroscopy include the analysis of food products, pharmaceuticals, combustion products, and a major branch of astronomical spectroscopy. In these instruments, the detector used for the NIR wavelengths is often the same detector used for the instrument's "main" range of interest. In the same way, the range of some mid-IR instruments may extend into the NIR. Many commercial instruments for UV/vis spectroscopy are capable of recording spectra in the NIR range (to perhaps ~900 nm). Multiple images may be recorded sequentially at different narrow wavelength bands. Instruments intended for chemical imaging in the NIR may use a 2D array detector with an acousto-optic tunable filter. Such instruments can record both UV-visible and NIR spectra 'simultaneously'. It is possible to combine silicon-based and InGaAs detectors in the same instrument. InGaAs and PbS devices are more suitable and have higher quantum efficiency for wavelengths above 1100 nm. Silicon-based CCDs are suitable for the shorter end of the NIR range, but are not sufficiently sensitive over most of the range (over 1000 nm). The type of detector used depends primarily on the range of wavelengths to be measured. When lasers are used, a single detector without any dispersive elements might be sufficient. For high precision spectroscopy, wavelength-scanned lasers and frequency combs have recently become powerful sources, albeit with sometimes longer acquisition timescales. Light-emitting diodes (LEDs) can also be used. Depending on the sample, the spectrum can be measured in either reflection or transmission.Ĭommon incandescent or quartz halogen light bulbs are most often used as broadband sources of near-infrared radiation for analytical applications. Fourier transform NIR instruments using an interferometer are also common, especially for wavelengths above ~1000 nm. There is a source, a detector, and a dispersive element (such as a prism, or, more commonly, a diffraction grating) to allow the intensity at different wavelengths to be recorded. Instrumentation for near-IR (NIR) spectroscopy is similar to instruments for the UV-visible and mid-IR ranges. It is only in the last few decades that NIRS began to be used as a medical tool for monitoring patients, with the first clinical application of so-called fNIRS in 1994. This optical method can be used in a number of fields of science including physics, physiology, or medicine.

With the introduction of light- fiber optics in the mid-1980s and the monochromator-detector developments in the early 1990s, NIRS became a more powerful tool for scientific research. In the 1980s, a single-unit, stand-alone NIRS system was made available, but the application of NIRS was focused more on chemical analysis. In the first applications, NIRS was used only as an add-on unit to other optical devices that used other wavelengths such as ultraviolet (UV), visible (Vis), or mid-infrared (MIR) spectrometers. The discovery of near-infrared energy is ascribed to William Herschel in the 19th century, but the first industrial application began in the 1950s. Near-infrared spectrum of liquid ethanol. Careful development of a set of calibration samples and application of multivariate calibration techniques is essential for near-infrared analytical methods. Multivariate (multiple variables) calibration techniques (e.g., principal components analysis, partial least squares, or artificial neural networks) are often employed to extract the desired chemical information. The molecular overtone and combination bands seen in the near-IR are typically very broad, leading to complex spectra it can be difficult to assign specific features to specific chemical components. Near-infrared spectroscopy is, therefore, not a particularly sensitive technique, but it can be very useful in probing bulk material with little or no sample preparation. One advantage is that NIR can typically penetrate much further into a sample than mid infrared radiation.
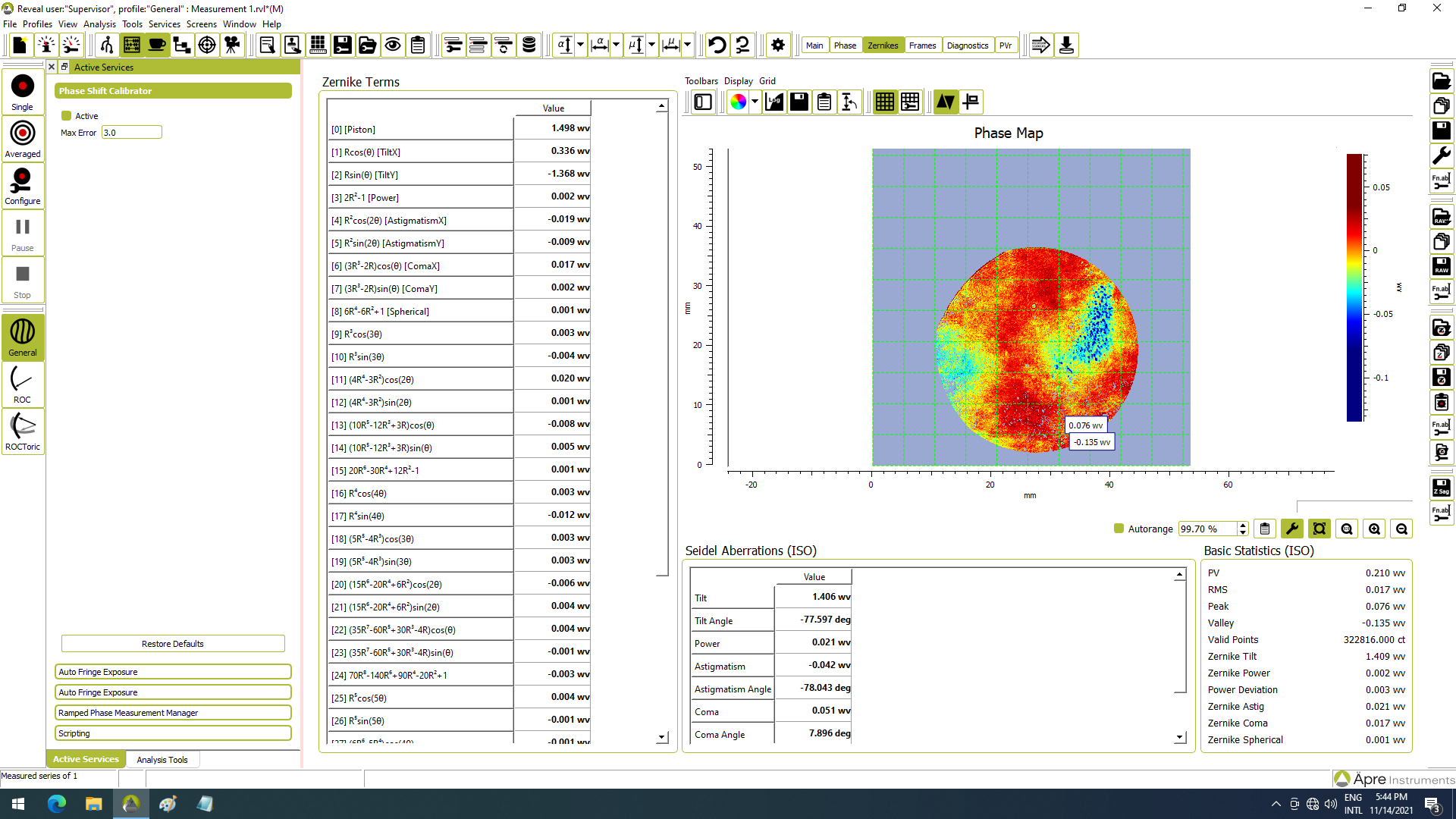
As a result, the molar absorptivity in the near-IR region is typically quite small.
Such transitions are forbidden by the selection rules of quantum mechanics. Near-infrared spectroscopy is based on molecular overtone and combination vibrations.
